Українською English
Main Scientific work Publications Visit card
Global scientific direction of the Group:
Design of Multi-channel Fluorescence Probes for the Study of Chemical Composition and Structure of Liquid Media
- Theory
- Design and Synthesis of Fluorescence Probes and Ion Indicators
- New classes of fluorescence dyes
1. Theory
In general, fluorescence probe is the molecular device, actually - a molecule that after absorption of light of optical range is capable to emit new light quantum of new wavelength. The wavelength (energy) of light in this case is dependent upon the properties of medium. By this way, due to special features of a structure that determine the physical and chemical properties, the molecule of probe has a possibility to transmit analytical information about the studied parameters of the object.
At present mono-channel (monoparametric) fluorescence probes have obtained wide applications in biology and medicine, since they are simple in use. Meantime only rare examples exist for design and applications of two- and three-channel probes.
 |
 |
 |
|
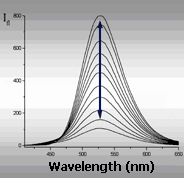
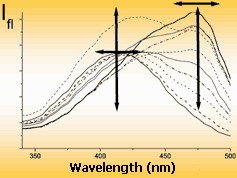
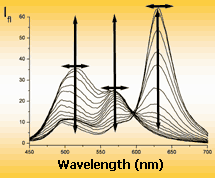
| Mono-channel | Excitation spectrum | Emission spectrum | |
| probe brings useful information in the change of single emission parameter only | Multi-channel (multiparametric) probe informs about his surroundings by the changes of several (to ten) emission parameters | ||
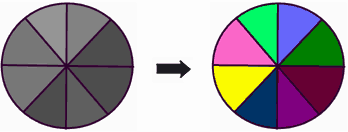
Mono-channel and multi-channel fluorescence probes on the efficiency of information transmission can be compared with black-and-white and color television. As for black-and-white television, main shortcoming of mono-parametric probe is the loose of important information about properties (about real color) of the object. Multi-channel probe has no such shortcomings. Example:
Black-and-white image of colored disc cannot display its real colors:
What are the principles of functions of multi-channel fluorescence probes? What structure and constitution they have? How to project and synthesize the fluorescence probe? Such questions should be answered during the creation of multi-channel fluorescence probes.
2. Design and Synthesis of Fluorescence Probes and Ion Indicators
 |
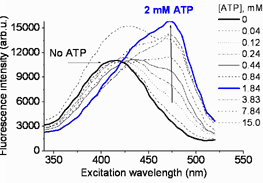 |
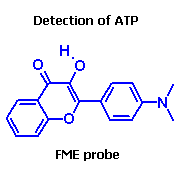
FME probe allows to detect adenosine triphosphate anion (ATP) in aqueous solutions in presence of ADP, GTP or other triphosphates. Range of detection: 0.05 - 5 mM. | |
|
V. G. Pivovarenko, O. B. Vadzyuk, S. O. Kosterin, J. Fluorescence. - 2006. - 16. - P. 9-15.
|
|||
|
Data group 1 obtained in MeOH-EtOAc mixtures, 2 – in MeOH-benzene, 3 – n-BuOH- benzene, 4 – t-BuOH- benzene ones. |
|
A method of determination of hydrogen bond donor molar concentration in a binary mixture was elaborated on this basis. It can be done by the four parameters of fluorescence spectrum of the probe: by the maxima positions of the tautomeric forms (νN*, νT*) and their peak intensities (IN*, IT*). Local polarity of probe microsurroundings can be evaluated simultaneously as a dielectric constant function (ε-1)/(2ε+1), since the last is in linear dependence with the sum of the positions of bands maxima νN*+νT*. |
|
Klymchenko S., Pivovarenko V. G., Demchenko A. P. Elimination of hydrogen bonding effect on the solvatochromism of 3-hydroxyflavones. J. Phys. Chem. A.- 2003.- 107.- P. 4211-4216. |
 |
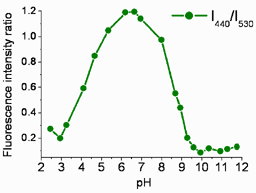 |
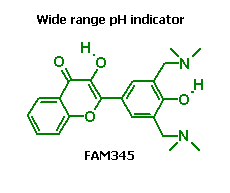
FAM345 indicator allows fluorometric determination of pH in range from pH 3 to pH 10. V.F. Valuk, G. Duportail, V.G. Pivovarenko. J. Photochem. Photobiol. A: Chem. 175 (2005) 226-231. |
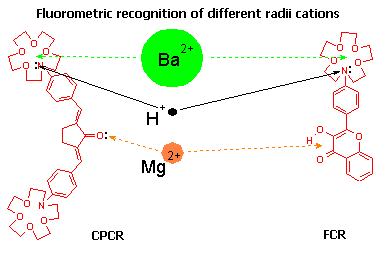 |
Fluorescence probes CPCR and FCRhaving two types of chelators give different response upon binding with cations of different size. These types of probes display the highest sensitivity in fluorescence to electrostatic effects in solutions (solvatochromism, electrochromism, etc.) |
|
|
A.O. Doroshenko, A.V. Grigorovich, E.A. Posokhov, V.G. Pivovarenko and A.P. Demchenko. Molecular Engineering 8 (1999) p.199-215.
|
||
 |
Studies of state and properties of lipid membranes with various flavonol probes: Phase transitions, surface and dipole potential investigations. Influence of HEPES buffer. Probes for apoptosis.
Bondar O.P., Pivovarenko V.G., Rowe E.S. Biochim. Biophys. Acta 1369 (1998) 119-30
|
3. New classes of fluorescence dyes
Diflavonols in the fluorescent probes design: intramolecular one or two proton transfer in the excited state. The most sensitive probes for polarity and hydrogen bond donating ability of the molecular surroundings. 3-Hydroxyquinolones (3HQ) - new class of excited state intramolecular proton transfer dyes with increased photostability.
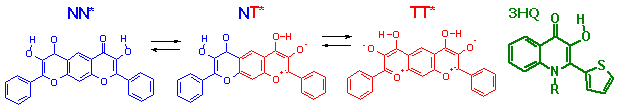
V.G. Pivovarenko, L. Jozwiak, and J. Blazejowski. Europ. J. Org. Chem, 2002, 3979-3985.
Falkovskaia E., Pivovarenko V.G., del Valle J.C.. J. Phys. Chem. A (2003) 107, 3316-3325.
Roshal A. D., Moroz V. I., Pivovarenko V. G., Wroblewska A., Blazejowski J. J. Org. Chem., 68 (2003) p. 5860-5869.
D. A. Yushchenko, M. D. Bilokin', O. V. Pyvovarenko, G. Duportail, Y. Mely, V. G. Pivovarenko. Tetrahedron Lett., 47 (2006) 905-908.
Dicyclopenta[b,e]pyridines: synthesis, conformations in solutions, response to pH.
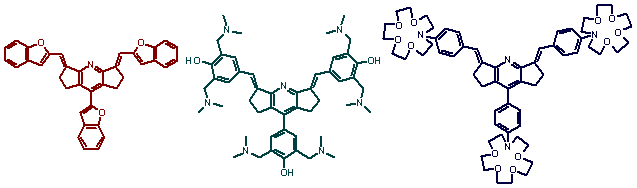
V.G. Pivovarenko, A. V. Grygorovych, V. F. Valuk, A. O. Doroshenko. J. Fluoresc., 13 (2003) 479-487.
V.F. Valuk, V.G. Pivovarenko, A.O. Doroshenko, A.V. Grygorovych. Teor Eksp. Khim., 40 (2004) 256-261.
V.F. Valuk, A.V. Grygorovych, A.O. Doroshenko, V.G. Pivovarenko. Ukr. Bioorg. Acta (2004) 1, 79-89.
Designed by Ukrainian2007